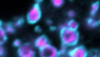
This high-resolution microscope image shows the donut-shaped outer mitochondrial membranes. Unlike typical mitochondrial transporters, SLC25A46 localizes to the outer mitochondrial membrane.
CORAL GABLES, Fla. (July 13, 2015)—Researchers at the University of Miami have discovered and characterized a previously unknown disease gene linked to the degeneration of optic and peripheral nerve fibers. Their study, “Mutations in SLC25A46, encoding a UGO1-like protein, cause an optic atrophy spectrum disorder,” is published in the journal Nature Genetics.
Patients with mutations in this gene present symptoms similar to optic atrophy and Charcot-Marie-Tooth Type 2 (CMT2), including vision loss and weakening of the lower leg and foot muscles beginning in the first decade of life.
The novel variants occur in a gene called SLC25A46 that functions in mitochondria, organelles inside animal cells known as the “cellular engines.” They transform food into fuel that allow cells to carry out energy-demanding functions.
“Mitochondria play a large role in human health,” said Alexander Abrams, Ph.D. student in neuroscience at the Miller School of Medicine and first author of the study. “Although we study rare diseases such as CMT2 and optic atrophy, the implications encompass all forms of neurodegeneration, including Lou Gehrig’s and Parkinson’s Diseases.”
Mitochondria constantly undergo fusion and fission to respond to cellular energy demands. By changing their size and connectivity through fusion and fission, mitochondria can travel to regions in cells where they are needed.
“Our study reveals that disrupting SLC25A46 causes mitochondria to become both more highly interconnected and improperly localized in cells,” said Julia E. Dallman, assistant professor of biology in the College of Arts and Sciences and a senior author of the study. “These data support a critical role for SLC25A46 and mitochondrial dynamics in the establishment and maintenance of neuronal processes.”
SLC25A46 encodes an atypical protein in the SLC25 family. SLC25 family members act like a channel, transporting molecules across the bilayer membranes inside mitochondria. But unlike the majority of human SLC25 family members (there are 53) that transport molecules across the inner mitochondrial membrane, SLC25A46 settles on the outer mitochondrial membrane where it regulates mitochondrial dynamics.
Mutations in the genes associated with mitochondria dynamics OPA1 and MFN2 are linked to similar mitochondrial disorders. Homologous genes in baker’s yeast work in combination with a gene called UGO1, which has ancestral similarities to SLC25A46. The new findings suggest that the SLC25A46 and UGO1 proteins may play similar roles.
Given the similarities between the diseases caused by mutations in OPA1, MFN2 and SLC25A46, these genes could be involved in common pathological mechanisms of neurodegeneration, the study says.
“This finding builds on our discovery of MFN2 as a major disease gene in this area over 10 years ago,” said Stephan Züchner, professor and chair of the Dr. John T. Macdonald Foundation Department of Human Genetics, at the Miller School of Medicine, and a senior author of the study. “Only through the new genome sequencing methods and active global data exchange were we able to solve this puzzle.”
The study is a collaborative effort with investigators from nine universities and research institutions in the United States, Italy, and the United Kingdom. Coauthors from UM’s Dr. John T. Macdonald Department of Human Genetics and the John P. Hussman Institute for Human Genomics are post-doctoral fellows Adriana Rebelo and Alleene V. Strickland, graduate Michael A. Gonzalez; Ph. D. candidate Feifei Tao, Fiorella Speziani, former research project manager; Lisa Abreu, clinical research coordinator; and Rebecca Schüle, M.D., Ph.D., visiting Marie-Curie Fellow. Other Miller School co-authors are Antonio Barrientos, professor of neurology, and Flavia Fontanesi, research assistant professor in the Department of Biochemistry and Molecular Biology.